Media
House Committee Releases Report on Humira and Imbruvica Price Increase Investigation
The U.S. House of Representatives Committee on Oversight and Reform released a 57 page report on May 18th, 2021 regarding AbbVie’s pricing of Humira and Imbruvica drugs in the United States.
AbbVie and Humira Pricing
Humira is a drug used to treat rheumatoid arthritis and other autoimmune diseases. It generated $160 billion in revenue for AbbVie in 2020 and has a current price of $77,000 per year. The price of the drug has increased nearly 5x over its initial pricing.
The Committee on Oversight and Reform reviewed 170,000 pages of internal documents from 2009 and to present, and concluded that the 470% “price increases [were] not justified”:
Internal data show that AbbVie’s list price increases for Humira and Imbruvica far outpaced any discounts and rebates paid to pharmacy benefit managers and other members of the supply chain. Humira’s net price—which subtracts discounts and rebates—increased by 110% between 2009 and 2018, from $16,663 per year to $35,041 per year.
The report finds that the result of the price increases was hundreds of millions of dollars in executive bonuses, which had been tied to Humira net revenue. The CEO eared $170 million in bonuses. The report noted that the largest price increases (30% in 10 months) occurred the first year executive compensation was tied to Humira revenue.
AbbVie and Imbruvica Pricing
Similar to Humira, Imbruvica prices increased substantially in a short period. From 2013 to 2017, the price for a year supply of the drug rose over 50%:
For a patient taking 3 tablets daily, the annual net price of Imbruvica increased from $72,587 in 2013 to $115,533 in 2017 (the last year for which AbbVie provided the Committee data).
Medicare Overpayment
As U.S. law does not allow Medicare from negotiating directly with drug companies, Medicare could not negotiate drug pricing with AbbVie. The report noted that had Medicare received the same discounting that the Department of Defense or Veterans Affairs received on drug orders, savings would have been $7 billion to $7.4 billion.
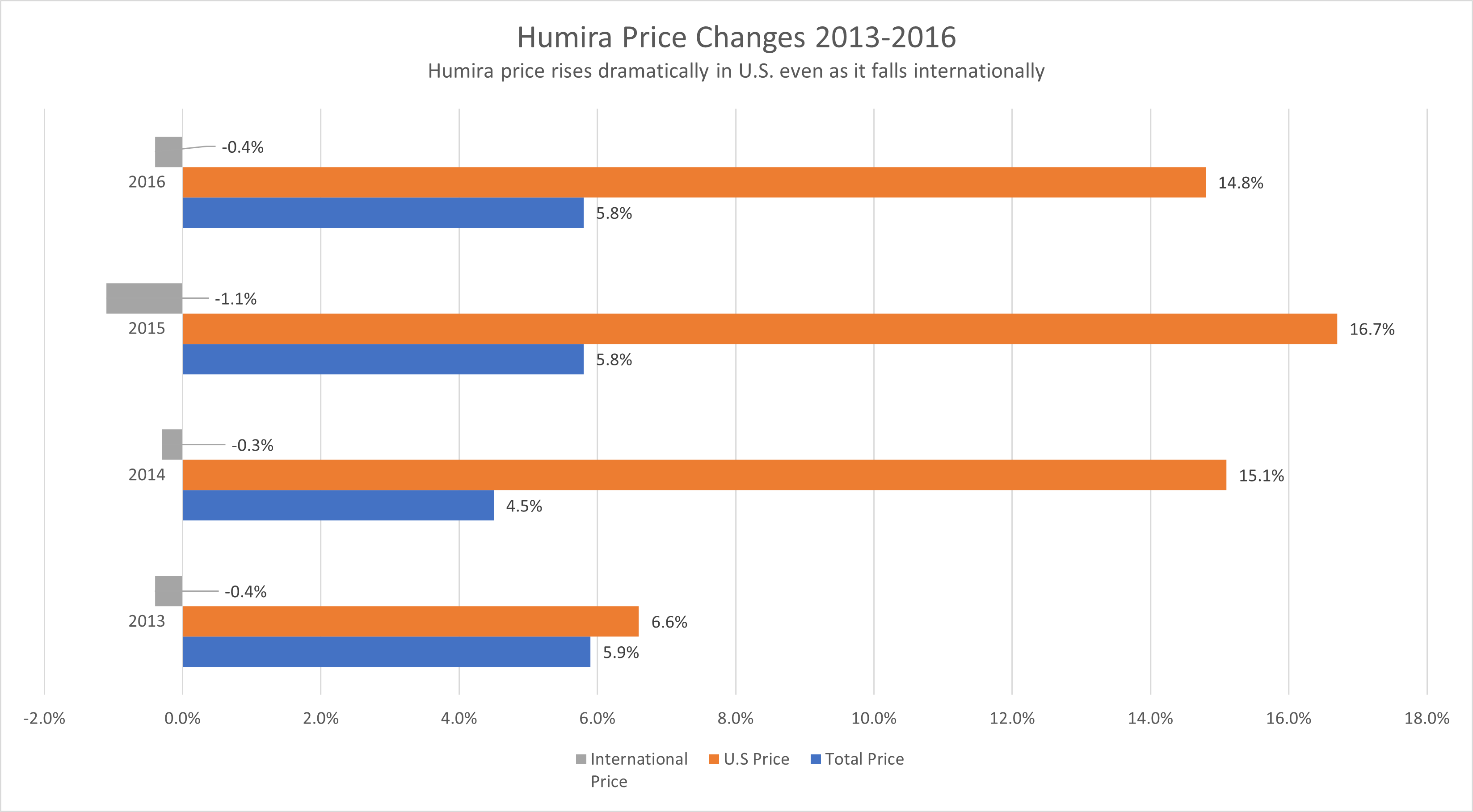
Humira and Imbruvica were sold to the US government and within the United States at inflated pricing not seen in other first world countries, including Canada, Japan, Korea and the United Kingdom. Pricing in the United States for both government and private purchasers rose according to the report even in times when prices fell internationally.
Patent Manipulation
The report accuses AbbVie of manipulating patent expiration by applying for over 250 separate patents on Humira to block similar drugs from the U.S. market. Patents for Humira do not full expire until the year 2037.
The report notes that the vast majority of patents were filed after Humira was on the market, and the government suggests this strongly shows the purpose of the patents was to slow competition and not protect or promote drug research and development. Imbruvica has over 150 patents which do not fully expire until 2036.
Pay-For-Delay Schemes, Alleged Abuse of Orphaned Drug Act
The report continues to accuse AbbVie of additional manipulation via pay-for-delay schemes for Humira. The company discovered multiple biosimilar drugs would enter the market as early as 2017, and AbbVie entered in agreements with four competitors to delay their market entry until 2023.
The question the report raises is whether AbbVie used monopoly profits from Humira to buy off competition illegally, effectively sharing the monopoly profits among potential competitors instead of competing in the market. The report estimates the lack of competition cost the U.S. government a total of $19 billion between 2016 and 2023.
The report makes additional allegation of abuse of the Orphan Drug Act, by applying for 8 “orphan” designations for a multi-billion dollar blockbuster drug in no danger of production problems. The Orphan Drug Act is intended to incentivize the development and production of new and existing drugs to treat rare conditions. AbbVie allegedly used the act to create different orphan protections and exclusively periods by dividing Humira use among different patients and age groups for the same diseases, for a drug with massive sales and production.
Price Fixing with Competitors
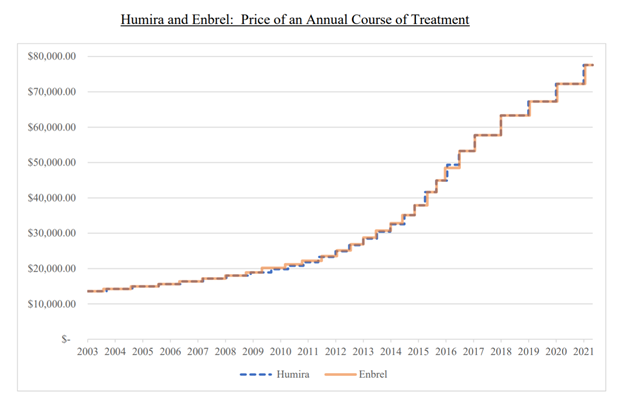
On top of all the allegations listed above, AbbVie is further alleged to have coordinated price increases with the few competitors not delayed from the market. The report shows that pricing between Humira and its closest competitor Enbrel from 2003 to 2021 shows coordinated movement:
Price Fixing Lawyers
Schneider Wallace has successfully represented insurance companies and other institutional plaintiffs in recovering for price fixing. Schedule an appointment with our legal team to learn more. Schneider Wallace has offices in California, Texas, North Carolina and Puerto Rico and litigates in jurisdictions throughout the country in state and federal administrative agencies and courts.
More information of Drug Price Fixing:
House Overnight Committee Reports on Drug Price Fixing, Schneider Wallace, Nov 2020